Learn how real-time AI documentation from Exactera could have saved $2.5M in at-risk R&D credits.
George v. Commissioner: Where It All Went Wrong
In today’s hyper-scrutinized tax environment, simply doing legitimate research and development is not enough to be able to claim R&D tax credits. A company must be able to prove it did legitimate research and development. Often, companies build the case for R&D tax credits retroactively, using a combination of existing data and employee interviews as proof for the work. But the case of George v. Commissioner shows what can go very wrong with this approach.
George’s of Missouri, Inc. (GOMI) — one of the largest fully integrated poultry processors in the U.S. — claimed $4.47 million in R&D tax credits across 2012–2014 for seven research trials on feed additives,vaccines, probiotics, and genetic lines. alliantgroup conducted the retroactive R&D study.
The Tax Court’s verdict was a blow. They disallowed three of seven trials, and the credit rate was slashed from 14% to 6%. Not because the research wasn’t real — but because the documentation couldn’t prove it.
GOMI’s research was legitimate — trials on Ross 708 genetic lines, probiotic additives like Sporulin and Calsporin, and vaccine protocols were real scientific work conducted by qualified personnel. But the R&D study was built retroactively by alliant group, relying on employee interviews conducted months or years after the research occurred. When the IRS compared those interviews narratives against GOMI’s own operational records — feed logs, flock data, procurement records — the contradictions were fatal.
The court found that feed records showed constant, unchanged dosages during periods where consultants claimed active experimentation was occurring. Entire flocks that allegedly received test additives had no documentation whatsoever. And credits were claimed for research years after the underlying uncertainty had already been resolved.
On top of the individual trial failures, GOMI couldn’t substantiate its base-year qualified research expenses, triggering a start-up limitation under IRC §41(c)(5)(B) that cut the credit rate from 14% to just6% — compounding the losses across every surviving claim.
THE DOCUMENTATION FAILURES THAT COST MILLIONS
Of seven research trials, three were completely disallowed and two were only partially allowed. Every failure traced to the same root cause: retroactive documentation that contradicted operational records.
● DISALLOWED / PARTIAL
Salinomycin & Phytase — Interview claims of experimentation contradicted by feed records showing constant, unchanged dosages (~$1.2M in QREs at risk)
Floramax — No records existed showing which flocks received the additive; the court refused to estimate (~$680K in QREs unsubstantiated)
HatchPak & Tylan — Credit claimed for 2013 after uncertainty was already resolved in 2012 ($2.87M credit year partially disallowed)
LT Vaccines — Priming trials allowed, but method-of-administration lacked timing records (partial credit reduction)
Combined with the base-year failure cutting the rate from 14% to 6%, the total credit impact was devastating across all $4.47M claimed
● ALLOWED — THE STANDARD THAT WON
Ross 708 Genetic Line — Called “the cleanest example of the scientific method” — control groups, data analysis, clear conclusions
Sporulin & Calsporin — Clear hypothesis, test, analyze, iterate pattern with documented next steps upon failure
These trials survived because their documentation matched what Exactera captures automatically for every project.
The Base-Year Penalty
Beyond individual trial failures, GOMI couldn’t substantiate base-year QREs (2009–2011). This triggered the start-up limitation under §41(c)(5)(B), slashing the credit rate from 14% to 6% — losing more than half the credit value on every qualifying trial. Exactera’s platform maintains rolling base-year data automatically, making this trap structurally impossible
WHY THIS KEEPS HAPPENING: THE RETROACTIVE STUDY MODEL
The failures in George aren’t unique — they’re structural. alliantgroup and traditional R&D firms all use the same retroactive model:
1. Research happens. No credit-specific documentation is captured.
2. 6–18 months later, consultants arrive and interview employees from memory.
3. Narratives are manually reconstructed — often contradicted by operational records the consultants never reviewed.
4. A static report is delivered. When the IRS finds discrepancies, there’s no way to fix them.
This is the model that produced three disallowed trials and a credit rate cut in half. The new Form 6765 makes this approach even riskier by requiring project-level detail and contemporaneous evidence that retroactive studies were never designed to produce.
HOW EXACTERA ELIMINATES THESE FAILURES

NEW FORM 6765 COMPLIANCE: THREE APPROACHES COMPARED
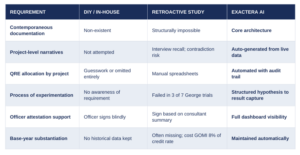
THE BOTTOM LINE
The George case isn’t ancient history — it was filed February 3, 2026. It proves that retroactive R&D studies produce documentation that crumbles under IRS scrutiny. Three trials disallowed. Credit rate cut in half. Millions in credits lost.
Every one of those failures traces back to the same root cause: documentation was reconstructed from memory instead of captured in real time.
Exactera’s AI platform doesn’t reconstruct the past. It documents the present. And that’s the difference between bcredits that survive an audit and credits that don’t.
Based on George v. Commissioner of Internal Revenue — T.C. Memo. 2026-10
This material is for informational purposes only and does not constitute legal or tax advice










